|
12/6/2023 0 Comments Checkmate 9la protocolAdditionally, the final analysis provided the results of PFS after the second line of treatment (PFS-2), defined as time from randomisation to progressive disease after next line of treatment or death. published the final analysis specified by the trial protocol, further confirming the significant benefit in terms of prolonging both OS and PFS ( 7). The initial results of the KEYNOTE-407 trial were published in 2018, demonstrating a longer overall survival (OS) and progression-free survival (PFS) in patients treated with the platinum doublet plus pembrolizumab ( 6). It should be noted that this trial excluded patients with symptomatic brain metastases or an ECOG of 2 or higher, which could limit its applicability to the real-world population. Patients received four cycles of the combination treatment, followed by a total of up to 35 cycles of pembrolizumab or placebo, provided there was no disease progression, death, or unacceptable toxicity. The chemotherapy regimen used in the trial was a combination of carboplatin and a taxane, either paclitaxel or nab-paclitaxel. The patient population was stratified based on PD-L1 tumour proportion score (TPS) of either less than 1% or greater than or equal to 1%, as determined by immunohistochemistry (IHC, Dako 22C3 antibody). KEYNOTE-407 was a phase III, randomized, global clinical trial comparing chemotherapy alone versus chemotherapy combined with pembrolizumab in patients with advanced squamous NSCLC. 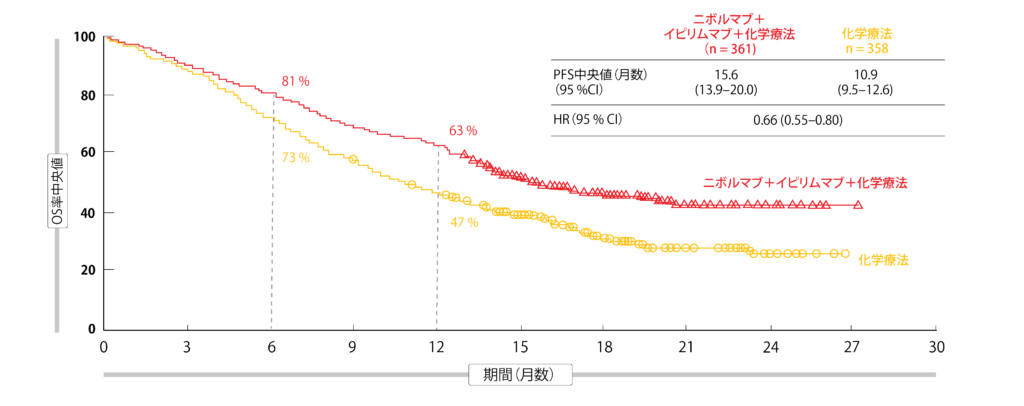
As the choice of chemotherapy regimen may vary based on histology, some trials were specifically tailored to evaluate these combinations in either squamous or non-squamous advanced NSCLC. 
Given these findings, several phase III trials were designed to compare chemotherapy plus ICI versus chemotherapy alone as first-line treatment for patients with metastatic NSCLC. However, ICI monotherapy did not demonstrate significant benefits in patients with PD-L1 expression levels below 50% ( 5). Initially, pembrolizumab was compared to platinum-based chemotherapy in patients with high (≥50%) PD-L1 expression, showing longer progression-free survival (PFS) and overall survival (OS) ( 4). The next milestone was the introduction of these drugs as first-line treatment options. Subsequently, atezolizumab and pembrolizumab were also shown to be more effective than docetaxel, leading to their approval for NSCLC treatment ( 2, 3). This approval was based on the results of a phase III clinical trial that demonstrated the superiority of nivolumab compared to docetaxel in this setting ( 1). In 2015, the Food and Drug Administration approved the first ICI, nivolumab, for patients with NSCLC whose disease had progressed after platinum-based chemotherapy. This advancement has significantly changed the therapeutic approach for NSCLC patients, providing new and more effective options for disease management. Over the past decade, the treatment landscape of metastatic non-oncogene addicted non-small cell lung cancer (NSCLC) has undergone a revolutionary transformation with the emergence of immune checkpoint inhibitors (ICIs), particularly those targeting the programmed death-1 (PD-1) axis. Keywords: KEYNOTE-407 pembrolizumab non-small cell lung cancer (NSCLC)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
